Atomatrix analyzed drug binding and signaling mechanisms targeting intractable cancers and hyperlipidemia, accelerating preparations for the launch of its drug design SaaS in 2026 | Image provided by Atomatrix
AI- and molecular dynamics (MD)-based drug discovery startup Atomatrix Co., Ltd. (CEO Eunho Lee) recently announced that it has gained international recognition for its technology by publishing two core research papers consecutively in Physical Chemistry Chemical Physics (PCCP), a leading global journal in the field of physical chemistry, just one year after its establishment.
The studies present results derived from applying key modules of Atomatrix’s in silico drug design platform, CANDDIE, to real disease targets. The platform’s BARon module predicts binding affinity, while Allopiper predicts pharmacological signaling pathways.
The first paper, titled “Mechanistic Insights into KRAS G12D Inhibitor Binding Revealed by Molecular Dynamics Simulations of Multiple Crystal Structures,” lists Dr. Donghwan Kim as first author. Using BARon-based molecular dynamics simulations, the study elucidates the binding mechanism of inhibitors targeting KRAS G12D, a mutation widely associated with intractable cancers. The research has attracted attention in academic circles for its potential to improve the precision of drug design for highly challenging cancer targets.
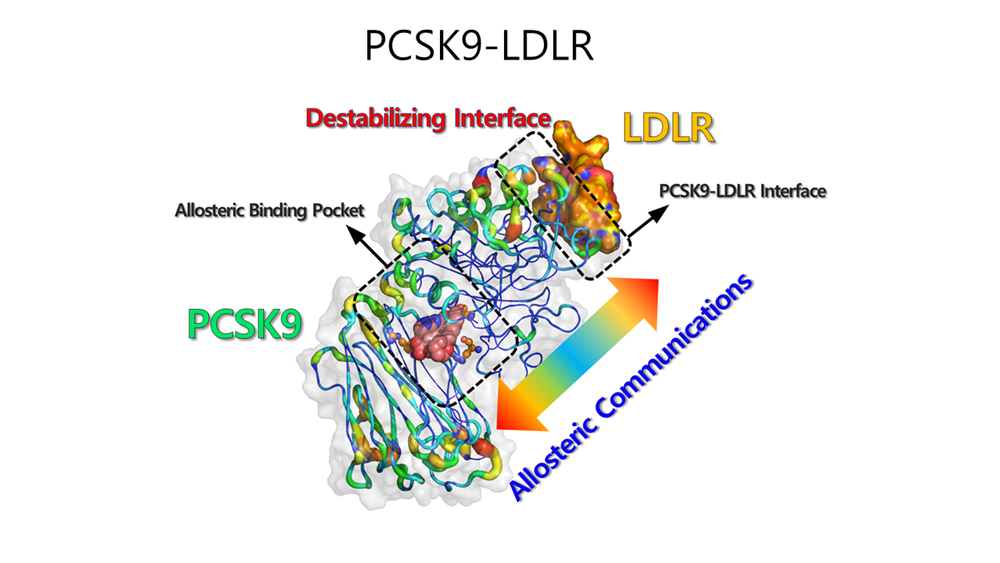
The second paper, “Allosteric inhibition of the PCSK9–LDLR interaction: structural insights for small molecule design,” lists Dr. Yong Su Baek as first author. Leveraging the Allopiper technology, the study reveals the allosteric regulatory mechanism of PCSK9, a key protein involved in cholesterol metabolism. The findings present a new structure-based design strategy for developing oral treatments for hyperlipidemia.
Since its establishment in May 2024, Atomatrix has achieved notable milestones within a year, including selection for the TIPS program and participation in the Seoul BioHub–Daewoong Pharmaceutical open innovation program, reflecting both its technological performance and growth potential. The company has also rapidly expanded its collaboration ecosystem by signing nine CDA agreements and two MOUs with domestic and global pharmaceutical companies.
Atomatrix unveiled the alpha version of CANDDIE at a nonclinical conference in Jeju earlier this year and announced plans to release a free beta version in March 2025, based on user feedback. The company is now accelerating the development of CANDDIE, an AI- and MD-integrated SaaS-type CADD platform, with an official launch targeted for June 2026.
Eunho Lee, CEO of Atomatrix, said, “Achieving both world-class academic results and industrial collaboration outcomes demonstrates the practical applicability of the CANDDIE platform,” adding, “We aim to create a new standard in drug discovery by intelligently connecting AI and experimentation.” Atomatrix plans to expand domestic and global partnerships further and advance data-driven drug design to enter the global bio software market in earnest.
